The Nature of Energy and Types of Energy
Energy—ability to do work or to supply heat. Mathematically,
where q = heat and w = work.
Heat—Thermal energy transferred from one object to another.
Another way of defining energy is by
where T = kinetic energy and V is the potential energy. The kinetic energy is the energy of motion for a particle with mass m, and has where v = velocity of particle. The potential energy is related to position of that particle. The functional form of potential energy differs in different systems. For example, we have seen in Chapter 7 that interaction energies between particles in H2 molecule is given by Coulomb interaction. The distances, hence position, between two charges give the strength of the interaction.
Energy conservation: The figure below explains the relationship between energy (total energy), kinetic energy, and potential energy. Let's say that you place a ball in a symmetrical bowl-like environment without friction. The ball has a mass, m. Since we are dealing with rolling a ball, we are dealing with the potential energy in gravity which has dependence on mass and the height of the ball. The potential energy is then given by
where g is the gravitational force on earth, 9.81 m/s2, and h is the height of the ball compared at the bottom of the well (h = 0). Energy unit is in Joules, J = 1 kg m2/s2. When you let the ball go, the position of where you put the ball, labeled with h0 = 1, the kinetic energy is zero at the instance of letting the ball go. The ball starts to accerelate until it passes the bottom of the well, labeled h1 = 0, at which the potential energy is zero, but kinetic energy is maximum, because of the pull of the gravity. The ball then starts to slow down until it reaches at the same height as h0 on the opposite side, labeled h2.
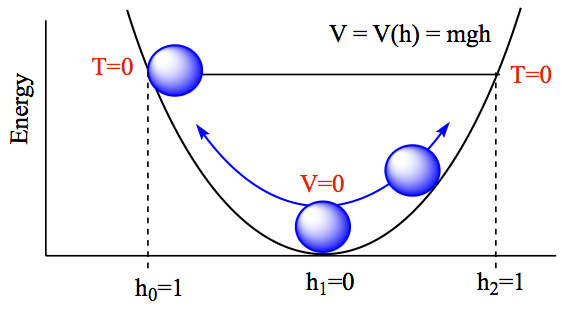
At each point of passage, the total energy is exactly the same.
- at h0, T = 0, E = V(h0),
- at h1, V = 0, E = T(h1),
- at h0, T = 0, E = V(h2),
- at any other position h, T ≠ 0, V ≠ 0, E = T + V
So, from these and in general, total energy of a process is conserved. Or total energy of the system is unchanged in a closed system.
Energy Change
Thermodynamic system: An isolated system, i.e. the system of interest such as moleucules being surrounded by surrounding, and the surrounding is enclosed by an insulated wall so that no heat, no work, no molecules escape from the surrounding. The system can exchange heat and/or work to the surrounding.
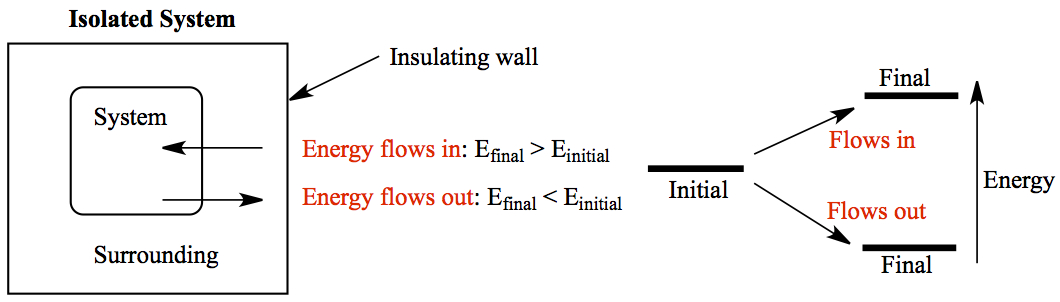
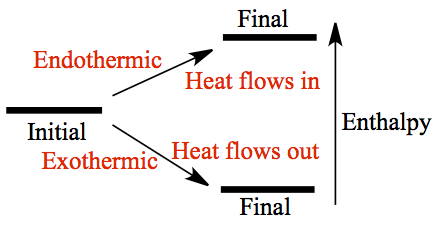
The energy of the system, internal energy which consists of molecular motions and interaction between them, can change so that the final energy can be higher or lower than its initial energy. When the final energy is higher than the initial, then the energy flows into the system from the surrounding. On the other hand, if the final energy is lower, the energy flows out of the system to the surrounding. These are depicted above.
State Function—if the quantity is independent of path, we call it state function. Energy is a state function. So, when calculating the overall energy change in the system, all you need to do, is to calculate the difference between the final energy and initial energy,
Introduction to Thermodynamics
We use the 1st law of thermodynamics and develop concepts that are useful in chemistry.
The difference between the final state energy and the initial state energy, called internal energy Work: In general work is force times distance. In this chapter, we only consider work involving expansion work. Under ordinary laboratory conditions, atmospheric pressure, p, is constant. So, when expansion work is performed by the system, work done by the system is given by the following equation,
where ΔV is the change in volume, and p is the pressure. Pressure is measured in atmosphere, atm, and the volume is measured in L with if pΔV = 1 atm L = 101 J.
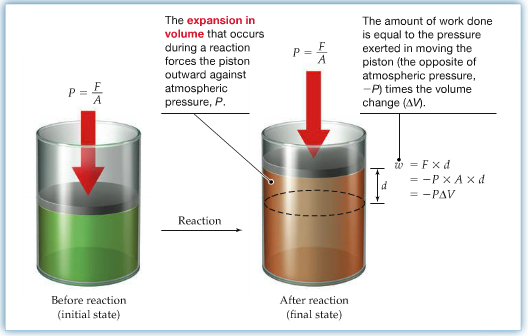
Since ΔV is defined as Vfinal - Vinitial, if the gas is expanded, meaning that Vfinal > Vinitial, the sign of the work is negative. In this case, it is said that the work is done by the system. If, however, the gas is compressed, then w is positive.
When the p-V work defined this way, and we can rewrite the change in the
energy of the system is,
If there is no work, that is no change in volume, q = ΔE.
If the pressure is constant, then q = ΔE + pΔV.
We define this heat measured at constant pressure to be
enthalpy change, ΔH.
Enthalpy is state function, like energy. So, enthalpy change is not depended on the path.
When the enthalpy of final state is higher than the initial state, it is said to be
endothermic and one must inject heat into the system to achieve the
final state. On the other hand, if the final state is lower in enthalpy, it is said to be
exothermic where heat is generated (or liberated) by the system.
Thermodynamic standard state—most stabel form of a substance at 1 atm pressure
and at specified temperature, usually 25°C. If solution is involved, the concentration
is 1 M.
We denote enthalpy change measure at the standard state to be ΔH° ← notice a
knot!
and
Then, by the virtue of enthalpy being state function, sublimation process is
Enthalpy change associated with chemical change Of course, we are
now talking about heat generated or absorbed by the reaction. We call
the heat, heat of reaction. We write down
the heat of reaction at the end of chemical equation, for example,
combustion of propane (C3H8)
The chemical equation that includes thermodynamic data, such as heat of
reaction, is called thermochemical equation. In the above example,
we have standard heat of reaction, and the reaction is exothermic.
If you reverse the reaction, that is,
the reaction becomes endothermic.
If on the other hand, the reaction is doubled the above,
If halved, the value of the ΔH° must be multiplied by ½.
The balanced equation is,
The rest is dimensional analysis, using 4.88g of barium hydroxide hexahyrate.
Since
The work term, according to ideal gas law, can be manipulated to
and if we assume temperature to be constant, then
Then,
Since the product produced one mole of gas, while reactant only has 0.5 mole
of gas, therefore Δn in the last term is Δn =
1.0 mol - 0.5 mol = 0.5 mol.
Since the enthalpy is calculated to be -110.5 kJ/mol,
The work term only contributes 1.2 kJ/mol or so. It depends on the temperature,
but generally the work term is quite small.
where ΔT = Tfinal - Tinitial.
If you want an equation, instead of proportionality, we can put a
proportionality constant, C, such that,
and C is called heat capacity.
Heat capacity tells how much energy required by a substance to raise its
temperature by 1°C. It has a unit of J/°C.
We can make heat capacity dependent on mass, it means that,
where m is the mass, and csp is
specific heat. Specific heat is defined as energy required
by 1 g of substance to raise its temperature by 1°C. Its unit is
J/(°C g).
Furthermore, we can make depended on number of moles, as well. In such a
case,
where n is the number of moles, and cm is the
molar heat capacity, which is defined as energy
required to by 1 mol of substance to raise its temperature by 1°C.
Its unit is J/(°C mol).
Using these, we can measure heat.
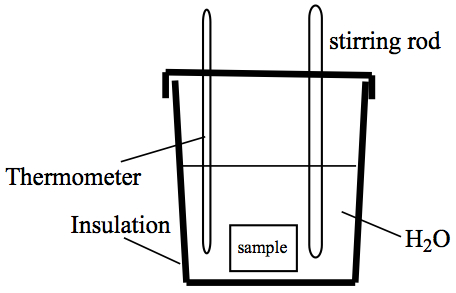
Calorimeter—a device to measure heat. Some devices are rather
elaborate, but the priciples are the same for any calorimeter. So, we
illustrate a calorimetry experiment using a styrofoam coffee cup, as follows.
The styrofoam cup acts as insulating walls in the description of isolated
system at the beginning of the chapter. We need to obtain temperature
readings, so a thermometer is necessary, and in order to keep the temperature
of water to be consistent throughout the cup, a stirring rod is provided.
A calorimeter usually use water as the surrounding to the sample chamber.
For simple experiments, such as the ones we perform in our labs,
it doesn't have to have the sample chamber.
Specific heat of water is given as 4.182 J/(°C g).
In elaborate experiments, we can not use specific heat of water as
the specific heat of surrounding, since the wall might absorb some of the
heat and it might change the value. The specific heat of the calorimeter
must be independently determined. For us in this course, we use the
specific heat of water as the specific heat of calorimeter.
If the system releases heat to the surrounding, the surrounding receives the
same amount of heat. So the amount of heat transferred is exactly the
same magnitude. However, in this case, system is "pushing" the heat, and
the surrounding is "pulling" the heat. This can be considered as opposite
effect. In terms of mathematics, we denote this "push-pull"
system to be,
where qsys is the heat from/to the system, and
qcal is the heat to/from the calorimeter (surrounding).
If we use the specific heat equation for q, then one can expand the
equation above as,
The ΔTsys on the left-hand side of the equation,
is given by Tfinal - Tinitial of the system,
while ΔTsys on the right-hand side of the equation,
is given by Tfinal - Tinitial of the calorimeter
or the surrounding. In either case, Tfinal is common
to both the system and the surrounding.
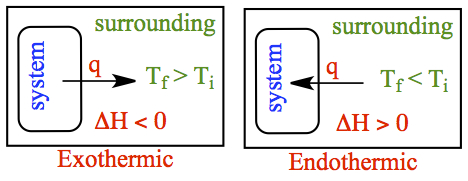
If the system is exothermic, then the heat is pushed into the surrounding.
Hence the temperature of the surrounding becomes higher, compared to the
initial temperature. On the other hand, if the system is endothermic,
the heat is extracted from the surrounding and provided to the system.
It means that the final temperature of the surrounding must become lower
than the initial temperature. These situations are shown in the figure to
the right.
In this case, the metal beads constitute a system, and the water is the
surrounding. So, we can use the push-pull system of heat.
where qmet is the heat of metal beads, and
qsur is the heat of surrounding (or water).
If we expand both sides, we get
Solving for specific heat of metal beads, we get
Another example is to solve for Tf.
ΔT is common to both water and metal, when it reaches
equilibrium. We can use the "push-pull" heat equation,
Expand this equation,
We can even apply the thermodynamic equation to ponder about the global climate
change. Here is an example that opens your eyes!
According to
Ohio State University's site the specific heat of air at 300 K is
1.005 kJ/kgK. According to
the Wiki page,
the mass of Earth's atmosphere is 5.15 x 1018 kg,
which was taken from the 1996 Edition of Handbook of Chemistry and
Physics.
We can calculate the energy available after 2°C increase.
What it means is that this energy is always available to the Earth, compared
to today.
Now, we should look at what this 1019 kJ of energy means.
We can look at, for example, the number of hurricanes we would see
with the amount of energy available due to the climate change.
According to NOAA,
the total energy released from cloud and rain is 5.2 x 1016kJ/day.
Let's say that a typical hurricane lasts 5 days. Then the total energy of one
hurricane is 2.6 x 1017kJ. According to
U of Arizona web site,
Hurricane Katarina produced 2.4 x 1017 kJ of energy.
The ratio between
the energy available by the climate change and the energy of typical
hurricane gives the number of hurricanes availble due
to temperature rise of 2°C, and it is 38. It means that the Earth would
carry, compared to today, 38 more hurricane equivalent energy always at
disposal. It is quite disturbing.
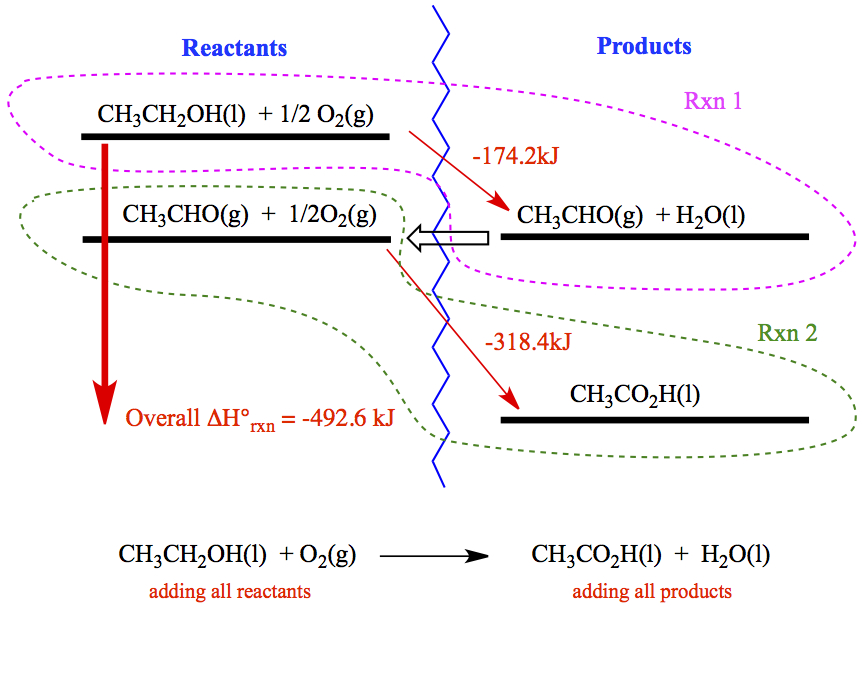
For example, we will look at the heat of reaction of the following.
When adding two equations We want the following equation as an
overall reaction, and ultimately want the ΔHrxn.
The two steps involved in this reaction are:
In addition, there is no C in the overall equation, therefore, we
must have the same number of C in both reactions. It means that
we need to multiply 2 to the first reaction, and 3 to the second.
Then, we have,
Here is an example of experiment we actually do in our lab dealing with the
Hess's law.
Rxn 2
MgO(s) + 2HCl(aq) →
MgCl2(aq) + H2O(l)
ΔH2 = ?
In addition to above two reactions, we need the heat of formation of water
to be able to make the overall equation to be correct.
Say that the following is the result of two reactions:
Heat of reaction 1 is calculated by by using,
Similar calculation for rxn 2 yields qcal = 2945.61 J.
Therefore, qrxn 1 = - qcal = -3636.56 J,
and qrxn 2 = - 2945.61 J.
Now we need to know the number of moles of Mg or MgO in the respective
solution so that q is converted to ΔH, and they are,
Dividing the qrxn 1 by the number of moles of Mg,
ΔHrxn 1 = -437.6 kJ/mol(Mg). Similarly,
ΔHrxn 2 = -114.7 kJ/mol(MgO).
Now we have to rearrange Rxn 1, Rxn 2, as well as the heat of formation of
H2O to obtain the equation of interest.
Rxn 1 stays the same since Mg appears on the reactant side in rxn 1 as
well as in the overall equation. However, rxn 2 needs to be reversed since
MgO appears in the product side of the overall equation,
meaning that multiply the rxn 2 equation by -1. Furthermore, we need to
cancel H2 in rxn 1, therefore, we need to divide the heats of
formation of water by 2.
Rxn 2
ΔHf
The enthalpy of formation of MgO is calculated to be -608.7 kJ/mol, which
is exothermic with large magnitude.
Hess's law can be illustrated in terms of energy diagram. For a reaction,
Rxn 2
CH3CHO(g) + ½O2(g)
→
CH3CO2H(l)
ΔH° = -318.4 kJ
as making liquid water from its elements, hydrogen molecule and
oxygen molecule. The enthalpy associated with the formation reaction
is called, heat of formation, ΔHf
. As we have seen already,
If we measure the heat of formation at standard state, then
the heat of formation is standard heat of formtion,
ΔH°f.
Heats of Formation of Elements are zero!
So, H2,
O2, C(graphite), Cu, Fe, Cl2 have all zero for its
formation enthalpy.
Standard Reaction Enthalpy. What's useful about the heats of
formation, is the fact that one can obtain heat of reaction, by the
following for the reaction,
where small letters, a, b, c, and d are the stoichiometry
of the balanced chemical equation. Then,
This is quite useful because you don't have to know anything about the
reaction to obtain the enthalpy of the reaction, simply by looking up
on a table of ΔH°f (In our case, look at
Appendix B in our text).
Chemical equation associated with the question is:
According to Table 6.4 of the textbook,
ΔH°f(CaCO3) = -1206.9 kJ/mol,
ΔH°f(CaO) = -635.6 kJ/mol, and
ΔH°f(CO2) = -393.5 kJ/mol.
Then, the
is given by,
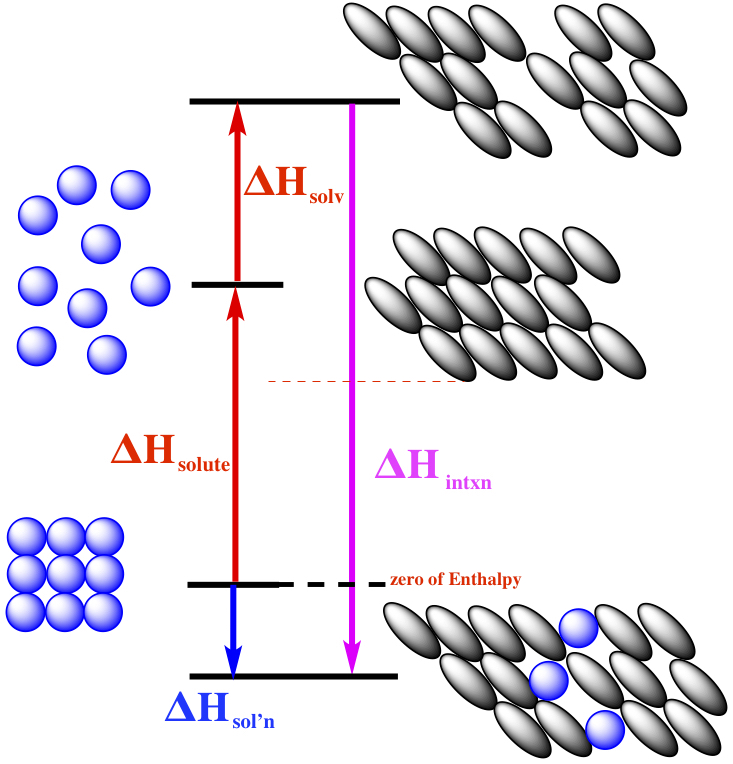
In the figure above, the heat associated with dissolving solute into
a solution is explained in terms of
three step process. The first step is to have the crystal (or any other
solid) with blue circles is separated into individual molecules. Since
there is attractive force exists between solute molecues, the process of
separation is endothermic, labeled as
ΔHsolute.
The solvent must make space for solute molecules to be embedded into the
solvent, so the heat needed to remove some of the solvent molecules to
make space for solute molecules again held by attractive forces,
is the ΔHsolv.
The heat associated with the
interaction of solute and solvent molecules are labeled as
ΔHintxn.
The overall process, ΔHsol'n,
is the blue arrow is the exothermic, in this particular case.
So, in order to dissolve some solute into some solvent we need to consider
three factors,
ΔHsolute,
ΔHsolv, and
ΔHintxn.
Enthalpy of Chemical Rxns
We already saw that ΔE = q - pΔV. Since we can measure heat, by
observing the temperatrue change, we can rearrange the equation and solve for q.
Then, we get,
Thermodynamic Standard State
It is convenient for us to use standardized thermodynamic quantity, so that when reporting
values can be compared directly.
Enthalpy of Physical and Chemical Change
Enthalpy change associated with physical change Melting process
(fusion), and boiling (vaporization) needs extra heat to melt or vaporize.
For H2O,
A comparison of ΔH and ΔU
In general, the internal energy change and enthalpy change have the similar
values. It tells you that the internal energy change is the dominant
quantity of the enthalpy chnage, and the work term is relatively small.
You can see that in the example below.
Calorimetry
Heat Capacity Since heat, q, has
to do with temperature change, heat and temperature change must be proportional
to each other, i.e.
Hess's Law
When multiple stpes are involved in the reaction, the sum of all enthalpy
changes in different stpes give the overall enthalpy of reaction.
overall eqn
2A + 2B → 3D ΔH = ?
rxn 1
A + B → 3C ΔH1
rxn 2
D → 2C
ΔH2
2(rxn 1)
2A + 2B → 6C
2ΔH1
-3(rxn 2)
6C → 3D
-3ΔH2
mass Volume Tinitial
Tfinal Rxn 1 0.202g (Mg) 100 mL HCl 21.7°C 31.7°C
Rxn 2 1.040g (MgO) 100 mL HCl 22.1°C 30.2°C
The concentration of HCl used in both cases is 2.87 M, and its specific
heat is 3.47 J/g°C. The density of HCl(aq) at the concentration is
1.048 g/mL.
2HCl(aq)
→
MgCl2(aq) + H2(g)
ΔH1 = -437.6 kJ/mol
MgCl2(aq) + H2O(l)
→
MgO(s) + 2HCl(aq)
ΔH2 = +114.7 kJ/mol
H2(g) + ½O2(g)
→
H2O(l)
ΔHf = -285.8 kJ/mol
Standard Heats of Formation
Formation Reaction—reaction to make a compound from
its stable elements. The reactants are the elements and the product is
the moleucle of interest. For example, the formation reaction for
water is,
Heat of Solution and Dilution
Heat of Solution
Heat being absorbed or released by the solute upon dissolving
to make a solution is called, enthalpy of solution, ΔHsol'n